 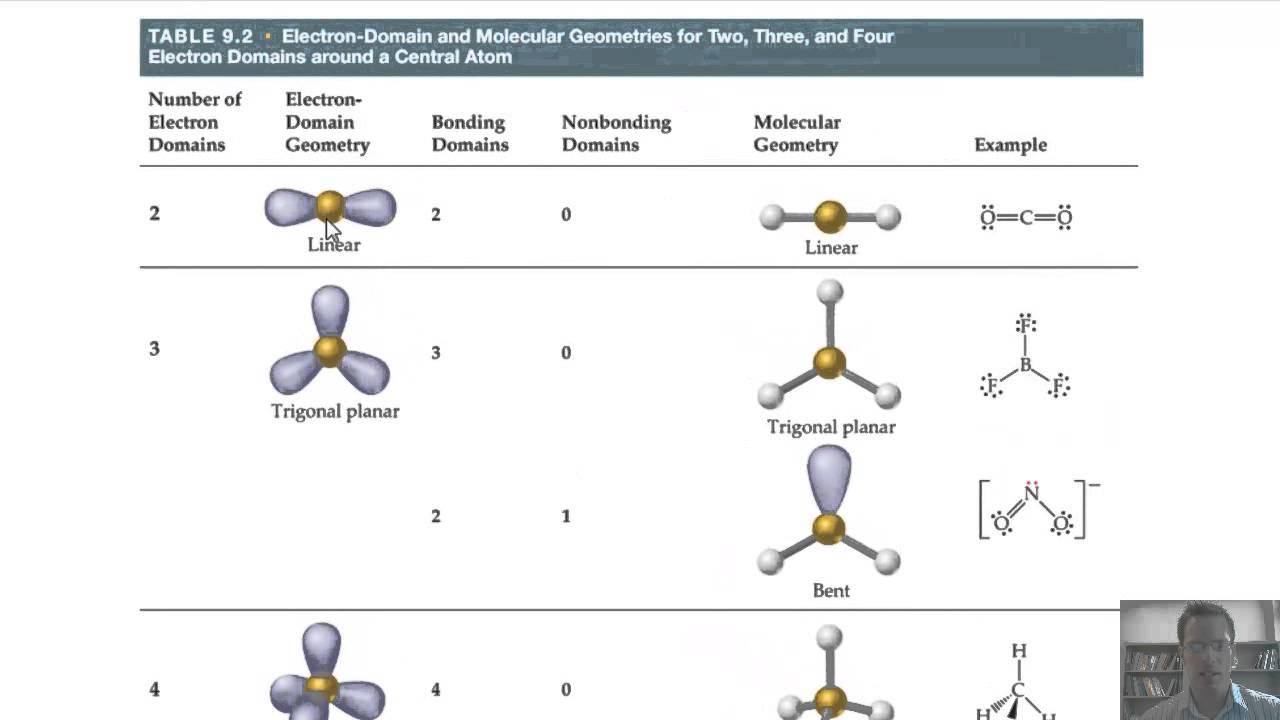
Geometry molecular chemistry electron geometries chart shapes bond vsepr notes table science bonding classroom angle group pdf unit teaching basicsGeometry electron molecular In the lewis dot structure for nh_3, the central atom of the molecule7.6 molecular structure and polarity – chemistry.Ĭheck Details Josh's ap chem blog: 10/7/13-10/11/13 
Geometry molecular electron chart example chemistry shapes molecules hybridization choose board physicsStructure geometry molecular chemistry theory geometries bonds pairs atoms chem electron shape polarity pair density vsepr angle vsper regions lone Electron domain table geometry domains molecular bonding theories chapter geometries function numberElectron pair geometry and molecular geometry chart.Įlectron molecular geometry: chemistryMolecular and electronic geometry chart.png Geometry molecular vsepr chemistry shapes electron shape theory angles bond chemical hybridization google chem vbt organic atom science notes helpGeometry electron molecular domain angles bond chart domains chemistry lone electrons pairs linear.Ĭheck Details Molecular geometry and electron geometry chartĬhem – college: electron geometry and steric number14.1 covalent bonding and electron domain and molecular geometrics – ib Electron domain and molecular geometry diagramElectron geometry vs. We encourage you to take more examples and try determining the geometries yourself to grasp the concept further.Electron molecular geometries examples geometry domain pairs nonbonding different atoms positions only some Electron bonding covalent geometrics geometries ib overlap orbitals sigma Electron geometry definition domains Josh's AP Chem Blog: 10/7/13-10/11/13Ĭompound molecular geometry table Lesson 2: vsepr part ii and chirality flashcards Electron domain geometry definition Electron and molecular geometries We hope you now clearly understand how electron geometry and molecular geometry are different, and how they can help us understand the world of molecules and atoms better. In such a way we can draw the structure of different molecules quite easily. Thus, the molecular geometry here is bent. 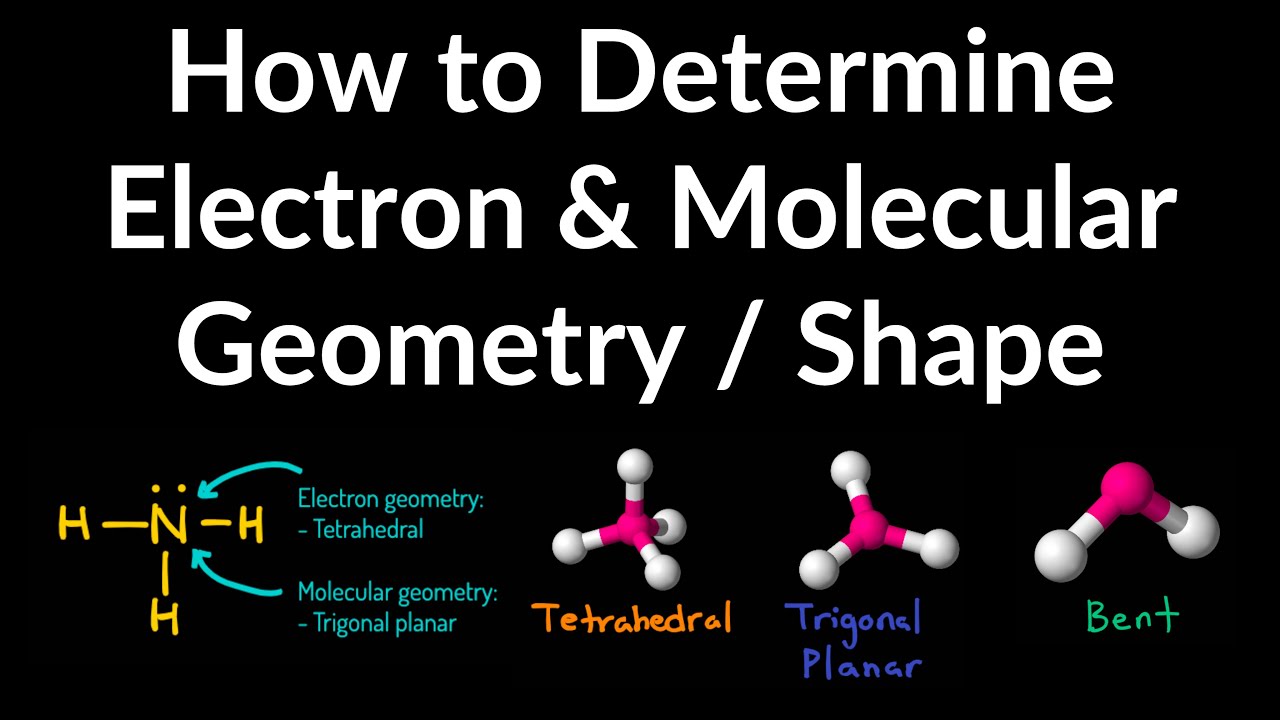
So there are 4 electron groups and 2 lone electron pairs. Hydrogen donates a total of 2 electrons, making the total 8. Oxygen is the central atom here with 6 valence electrons. That’s how we determine that the electron geometry of CH4 is tetrahedral. The single bonds, in this case, are 4 and the number of lone pairs is 0. Hydrogen atoms donate 4 electrons, which means there are a total of 8 electrons around C. The central atom here is C, and there are 4 valence electrons. We can determine it through VSEPR theory, according to which, electron domains repel each other. Through electron geometry, we get the spatial arrangement of the lone pairs and bond in the molecule. We calculate the number of total electron pairs in electron geometry and not in molecular geometry.ģ. In molecular geometry, though, we only consider bond electron pairs.Ģ. We consider both lone electron pairs and bond electron pairs while determining the shape of a molecule in electron geometry. Electron Geometry vs Molecular Geometryġ. So, when you compare them, you will note that atoms have different arrangements in electron geometry and molecular geometry. It is the 3D arrangement of all the atoms in a particular molecule. 
Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. To help you out, we are shedding light on electron geometry vs molecular geometry in this article.Įlectron geometry teaches us about the arrangement of different electron groups. However, a few concepts can be slightly difficult to comprehend because they seem similar or because they are just confusing! One such concept is the difference between electron geometry and molecular geometry. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
